Tel:0086-21-58356513, 58356613
Email:info@skybluechem.com
Site:www.skybluechem.com/
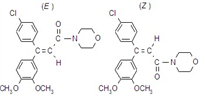
Common name dimethomorph (BSI, draft E-ISO); diméthomorphe ((m) draft F-ISO)
IUPAC name (E,Z)-4-[3-(4-chlorophenyl)-3-(3,4-dimethoxyphenyl)acryloyl]morpholine
Chemical Abstracts name (E,Z)-4-[3-(4-chlorophenyl)-3-(3,4-dimethoxyphenyl)-1-oxo-2-propenyl]morpholine
CAS RN [110488-70-5]
PHYSICAL CHEMISTRY
Composition (E)- to (Z)- ratio is c. 1:1. Mol. wt. 387.9 M.f. C21H22ClNO4 Form Colourless crystals. M.p. 127-148 ºC; (E)- isomer 135.7-137.5 ºC; (Z)- isomer 169.2-170.2 ºC V.p. (E)- isomer 9.7 ´ 10-4 mPa; (Z)- isomer 1.0 ´ 10-3 mPa (both 25 ºC) KOW logP = 2.63 (E)- isomer; 2.73 (Z)- isomer (both 20 ºC) S.g./density Bulk density 1318 kg/m3 (20 ºC) Solubility In water 19 (pH 5), 18 (pH 7), 16 (pH 9) (all in mg/l, 20 °C). In acetone 88 (E), 15 (Z), cyclohexanone 27 (Z), dichloromethane 315 (Z), dimethylformamide 272 (E), 40 (Z), hexane 0.04 (E), 0.02 (Z), methanol 7 (Z), toluene 7 (Z) (all in g/l, 20-23 ºC). In n-hexane 0.11, methanol 39, ethyl acetate 48.3, toluene 49.5, acetone 100, dichloromethane 461 (all for (EZ), in mg/l). Stability Hydrolytically and thermally stable under normal conditions. Stable for >5 years in the dark. The (E)- and (Z)- isomers are interconverted in sunlight.
APPLICATIONS
Biochemistry Inhibits the formation of the oomycete fungal cell wall.
Mode of action Local systemic fungicide with good protectant and antisporulant activity. Only the (Z)- isomer is intrinsically active, but, because of rapid interconversion of isomers in the light, it has no advantage over the (E)- isomer in practice.
Uses Fungicide effective against Oomycetes, especially Peronosporaceae and Phytophthora spp. (but not Pythium spp.) in vines, potatoes, tomatoes and other crops. Used in combination with contact fungicides, and applied at 2.0-2.5 kg dimethomorph/ha.