Tel:0086-21-58356513, 58356613
Email:info@skybluechem.com
Site:www.skybluechem.com/
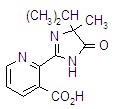
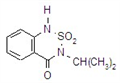
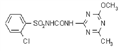
imazethapyr
Common name imazethapyr (BSI, ANSI, draft E-ISO, (m) draft F-ISO)
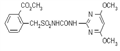
IUPAC name (RS)-5-ethyl-2-(4-isopropyl-4-methyl-5-oxo-2-imidazolin-2-yl)nicotinic acid
Chemical Abstracts name (?-2-[4,5-dihydro-4-methyl-4-(1-methylethyl)-5-oxo-1H-imidazol-2-yl]-5-ethyl-3-pyridinecarboxylic acid
CAS RN [81335-77-5]
PHYSICAL CHEMISTRY
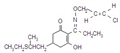
imazethapyr
Mol. wt. 289.3 M.f. C15H19N3O3 Form Colourless crystals. M.p. 169-173 ºC B.p. Decomp. 180 °C V.p. <0.013 mPa (60 ºC) KOW logP = 1.04 (pH 5), 1.49 (pH 7), 1.20 (pH 9) (all 25 ºC) S.g./density 1.10-1.12 (21 °C) Solubility In water 1.4 g/l (25 ºC). In acetone 48.2, methanol 105, toluene 5, dichloromethane 185, dimethyl sulfoxide 422, isopropanol 17, heptane 0.9 (all in g/l, 25 ºC). Stability Rapidly degraded in sunlight, DT50 c. 3 d. pKa pKa1 2.1, pKa2 3.9
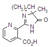
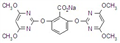
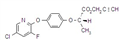
imazethapyr-ammonium
Mol. wt. 306.4 M.f. C15H22N4O3
APPLICATIONS
Biochemistry Branched chain amino acid synthesis (ALS or AHAS) inhibitor. Hence reduces levels of valine, leucine and isoleucine, leading to disruption of protein and DNA synthesis. Selectivity in soya benas and peanuts is attributed to rapid detoxification via hydroxylation and glycosylation (B. Tecle et al., Proc. 1997 Br. Crop Prot. Conf. - Weeds, 1, 605).
Mode of action Systemic herbicide, absorbed by the roots and foliage, with translocation in the xylem and phloem, and accumulation in the meristematic regions.
Uses Control of many major annual and perennial grass and broad-leaved weeds in soya beans and other leguminous crops. Applied pre-plant incorporated, pre-emergence, or post-emergence.
Phytotoxicity Non-phytotoxic to soya beans and other leguminous crops, when used as directed.